Sponsorship & Exhibits
ICNMD is an excellent opportunity to interact with a diverse group of clinicians, scientists, students, and industry professionals involved with muscular dystrophies, other myopathies, myasthenia gravis, polyneuropathies, spinal cord disorders, and neurofibromatosis, to list only a few. Over 1,000 clinicians and scientists from around the world are expected to attend.
Increase your visibility and organization’s exposure to national and international markets by becoming a sponsor and/or exhibitor or by holding an industry-supported symposium at this outstanding and important Congress.
Please see below for background information and for the link to request our sponsorship brochure.
The Congress is pleased to offer a variety of options, including but not limited to:
- Industry-supported seminars
- Print, digital and onsite advertising opportunities
- Exhibit booths
- and more…
To receive more details on how to sponsor or exhibit at our event, please fill out our Brochure Request Form.
 Important: This form is NOT for financial assistance or registration support for participants. We do not offer funding for delegates to attend the Congress.
Important: This form is NOT for financial assistance or registration support for participants. We do not offer funding for delegates to attend the Congress. Statistics on Current and Past Editions
Event Attendance
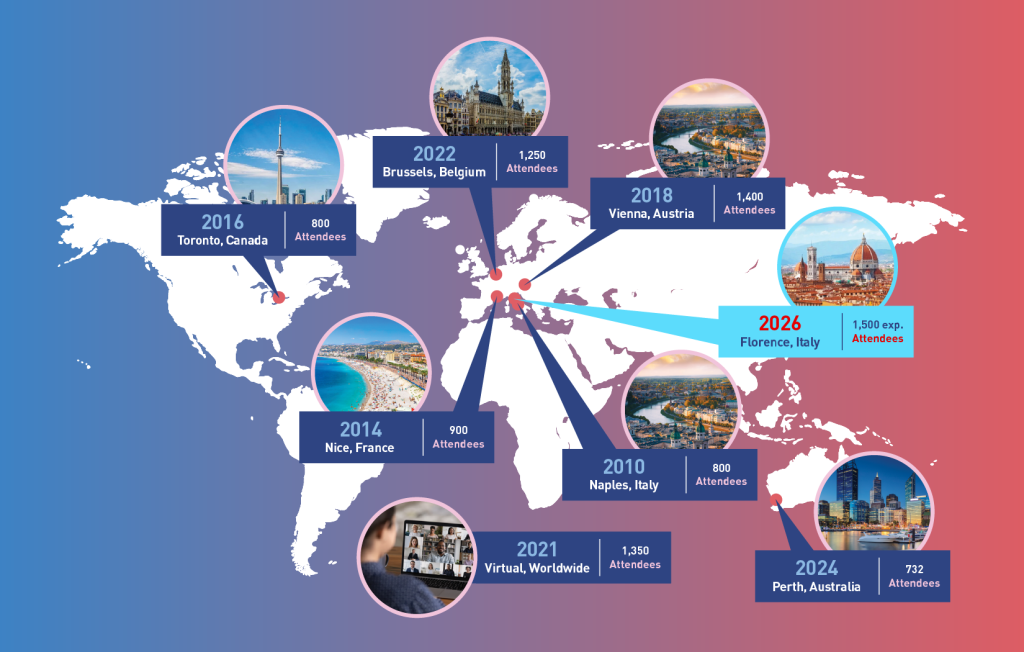
Demographics & Statistics
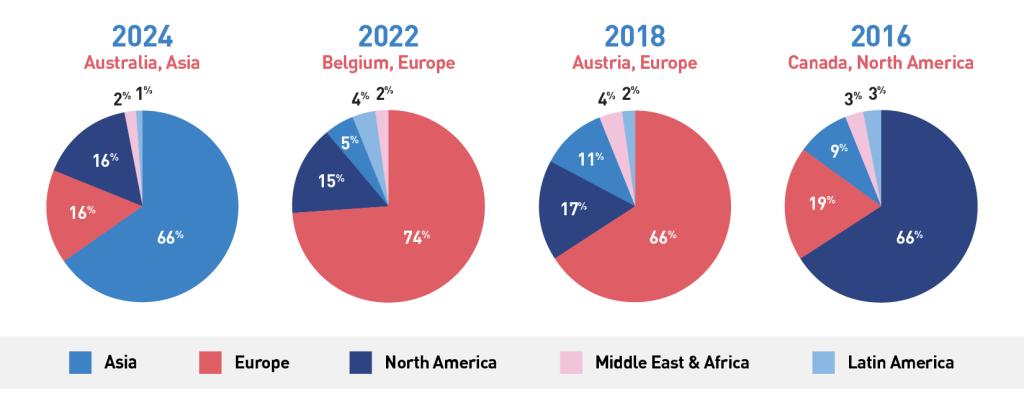
Regional statistics from past ICNMD editions:
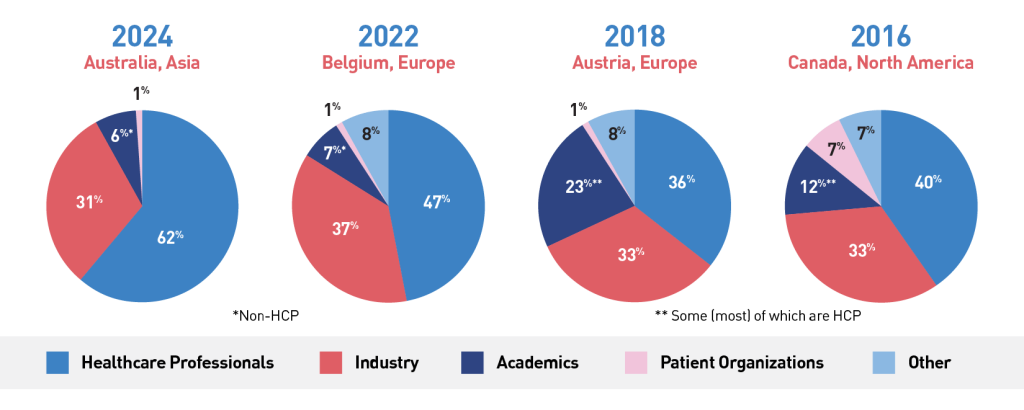
Category statistics from past ICNMD editions:
Past Sponsors & Exhibitors
|
|
Ancillary Meetings
Any meeting organized in conjunction with ICNMD by industry with participants who are registered Congress attendees (regardless of professional role or designation) will be deemed an Ancillary Meeting, whether held onsite or offsite.
Examples include roundtable discussions, steering committee meetings, advisory board meetings, investigator meetings, meet-the-specialist meetings.
Meetings that do not fall under this category are internal/staff meetings where only company staff are participating (whether attending the Congress or not).
Ancillary Meetings are not allowed to take place during blackout times, so as not to conflict with the official scientific program.
Blackout Times:
| Tuesday 7 July | 17:15 - 19:30 |
| Wednesday 8 July | 08:30 - 18:15 |
| Thursday 9 July | 07:00 - 18:15 |
| Friday 10 July | 07:00 - 22:00 |
| Saturday 11 July | 08:30 - 16:15 |
All times are subject to change.
Meetings with fewer than 10 attendees and held onsite in a space reserved through ICNMD may take place at any time during the Congress. They are not subject to the above Blackout Times. They are still subject to approval and fee.
All Ancillary Meetings, whether held onsite or offsite, must be submitted to, and approved by, the Congress Secretariat.
Please provide Day, Time, Location, Audience Numbers, Audience Type, and Topic.
See Contact Us paragraph below for contact information.
A Meeting Fee (space rental fee) is required for meetings held onsite. Duration includes setup, slide prep, meeting time, and teardown.
| EUR 6,000 | 1 day (8 hrs) |
| EUR 3,000 | ½ day (4 hrs) |
| EUR 1,000 | 1 hr |
This fee includes a basic AV package, including projector and screen. Requests or modifications outside this basic package may incur additional fees. See Contact Us paragraph below for contact information.
Supporters found hosting an onsite or offsite Ancillary Meeting without prior approval by the Congress Secretariat will be subject to a penalty of EUR 15,000 and/or removal of benefits.
Requirements with the Italian Medicines Agency (AIFA)
As required by current regulations of the Italian Medicines Agency (AIFA), all companies holding marketing authorizations (MAs) in Italy or with registered offices, branches, or representative offices in Italy who intend to contribute financially or through sponsorship to educational events involving drugs for human use are required to submit a pre-request for authorization to AIFA at least 70 days prior to the event.
Please note that the AIFA authorization is *NOT* necessary when:
- A company promotes only medical devices or food supplements during a meeting (products without Marketing Authorization) but subjected to MEDTECH authorization (if the company is associated);
- When a company sponsors a meeting about arguments not related to the use of any of its pharmaceutical products; in this case the company is not allowed to expose or distribute any kind of advertising material during the meeting (Section 9 art. 124 D.L. n.219/06).
Fargo International di Giulia Cambria
Via Pietro Maroncelli, 32
50137 Firenze
Italy
P.Iva 07325650484
Email: info@fargointernational.it
Ref. Giulia Cambria
Contact Us
Please contact our dedicated Partnerships Manager for more information on our sponsorship and exhibits options, and to start discussing how you would like to support and be involved in ICNMD.
Mrs. Cali (Carine) Vindeirinho, B.Eng., M.Sc.
Senior Manager, Partnerships
ICNMD-Sponsorship(at)icsevents.com
Direct: [+1] 604 639 3919 (Pacific Standard/Daylight Time)
Office: [+1] 604 681 2153 x 138
